Home / Explore Insights / Estimands: An insight into the new framework
/ Insights / Explore Insights / Estimands An Insight Into The New Framework
Estimands: An insight into the new framework
The objective of a clinical trial is to answer key clinical questions. Defining how the trial objectives are going to be estimated involves several key pieces of information such as which subjects to include, how to handle missing data, and the analysis method. An estimand framework has been presented by the ICH in November 2019 and the FDA in May 2021 to provide clarity on defining the reported treatment effect. This framework should be utilized from the initial trial planning stages and allows transparency for both sponsor and regulatory reviewers into how the study treatment effect will be presented at the end of the trial. Below summarizes the key components to the estimand framework that are covered in the ICH E9 (Revision 1) addendum on estimands and sensitivity analyses in clinical trials, as well as the supporting FDA guidance and ICH training material:
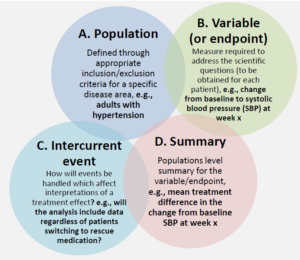
An estimand is characterized by four attributes to address the clinical question:
- The targeted study population
- The variable or endpoint to be obtained for each patient
- Information on how intercurrent event(s) will be handled, where an intercurrent event is defined as events that occur after treatment initiation and either preclude observation of the variable or affect its interpretation
- Population-level summary for the variable of interest, which provides a basis for comparison between treatment conditions
An example of how these four attributes can be used is shown below:
Estimand (given all 4 attributes) could be written as: Mean change from baseline in SBP of week x in the treatment group versus a control group regardless of whether rescue medication given, in adult patients with hypertension meeting the inclusion/exclusion criteria of the study.
The ICH E9(R1) addendum also clarifies the difference between a sensitivity analysis and a supplementary analysis—two concepts that are often interchanged despite different statistical meanings:
-A sensitivity analysis is conducted with the intent to explore the robustness of inferences from the main estimator (i.e. the method of calculating the estimate). An example could be running the analysis using a different imputation method for missing data than the primary estimand.
-A supplementary analysis is conducted in addition to the main and sensitivity analysis, to provide additional insights. Therefore, supplementary analyses are broader than sensitivity analyses. An example could be conducting the same analysis but on a different analysis set, such as the modified intent-to-treat set.
A fellow statistician once stated: think of an Estimand as ‘a mindsEt’ (notice the anagram). That is because a key goal of this new framework is to create a collaborative mindset between clinicians, statisticians, and ultimately the regulators and markets that will approve and buy new drugs. That is how we will better phrase our clinical trial objectives and answer them in a way that all key stakeholders will understand precisely what was estimated.
Rho has experience constructing estimands in both Protocols and Statistical Analysis Plans (SAPs).