Experience, a Collaborative Partnership
UNITY and Rho’s Answer to Mitigating COVID-19 Risk in a Complex, Fast-Moving Osteoarthritis Program
UNITY Biotechnology partnered with us to run a complex and fast-moving Osteoarthritis program with three active, concurrent studies across 25 sites and 218 patients when COVID-19 hit. Interim database locks were already scheduled four months away with final database lock deadlines looming close behind. The pandemic put that all on the line.
The UNITY and Rho teams had to pivot quickly to find creative solutions for site availability, patient retention, and remote monitoring that would ensure patient safety, data quality, and allow study progression to continue without delay – while following pandemic-related guidelines. Using a customized and decentralized multi-layered strategy, UNITY and Rho were able to successfully transition all three active studies in under four months, maintain integrity and ultimately meet critical database lock timelines.
3 studies were successfully transitioned in under 4 months, while maintaining pre-pandemic metrics for:
- Patient retention
- Recruitment and overall program timeline
- Database lock
Rho has been an exceedingly and consistently reliable partner in our three pain centric clinical trials in OA, and a true extension of our team at UNITY.
Associate Director, Clinical Operations, UNITY Biotechnology
Hear from both UNITY Biotechnology and Rho about the creative and collaborative solutions they implemented to pivot quickly and meet critical study timelines for a complex and fast-moving Osteoarthritis program – all in the midst of a pandemic.
Featured Content
Rho’s Answer to Mitigating COVID-19 Risks in a Complex, Fast-Moving Pain Program
Take a journey through the challenges, creative approach, and real-world outcomes of UNITY and Rho’s collaborative partnership to keep three Osteoarthritis studies on track during COVID-19.
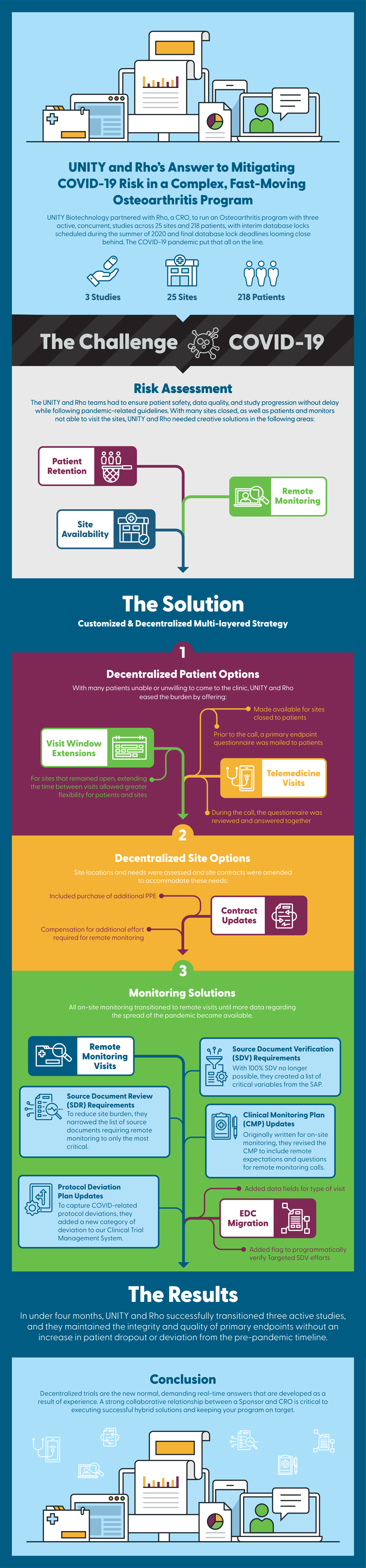
Decentralized trials are the new normal, demanding real-time answers that are developed as a result of experience, and a willingness to adjust and adapt to any situation. Contact our experts to learn more about how a collaborative relationship between Sponsor and CRO is critical to tackling challenges and keeping your program on track.

