Home / Explore Insights / FDA Guidance on Non-Inferiority Clinical Trials to Establish Effectiveness
/ Insights / Explore Insights / FDA Guidance on Non-Inferiority Clinical Trials to Establish Effectiveness
FDA Guidance on Non-Inferiority Clinical Trials to Establish Effectiveness
Heather Kopetskie, MS, is a Senior Biostatistician at Rho. She has over 10 years of experience in statistical planning, analysis, and reporting for Phase 1, 2 and 3 clinical trials and observational studies. Her research experience includes over 8 years focusing on solid organ and cell transplantation through work on the Immune Tolerance Network (ITN) and Clinical Trials in Organ Transplantation (CTOT) project. In addition, Heather serves as Rho’s biostatistics operational service leader, an internal expert sharing biostatistical industry trends, best practices, processes and training.
In November 2016, the FDA released final guidance on Non-Inferiority Clinical Trials to Establish Effectiveness providing researchers guidance on when to use non-inferiority trials to demonstrate effectiveness along with how to choose the non-inferiority margin, test the non-inferiority hypothesis, and provide interpretable results. The guidance does not provide recommendations for how to evaluate the safety of a drug using a non-inferiority trial design. This article provides background on a non-inferiority trial design along with assumptions and advantages and disadvantages of the trial design.
Background
A non-inferiority trial is used to demonstrate a test drug is not clinically worse than an active treatment (active control) by more than a pre-specified margin (non-inferiority margin). There is no placebo arm in non-inferiority trials. A non-inferiority trial design is chosen when using a placebo arm would not be ethical because an available treatment provides an important benefit, especially for irreversible conditions (e.g. death). Without a placebo arm to compare either the test or active control against it is important to determine that the active control had its expected effect in the non-inferiority trial. If the active control had no effect in the non-inferiority trial it would not provide evidence that the test drug was effective.
The table below compares superiority with non-inferiority trials with respect to the objective and hypotheses. The effect of the test drug is ‘T’ and the effect of the active control is ‘C’. The difference tested during analyses is C – T.
| Superiority Trial | Non-inferiority Trial | |
| Objective | To determine if one intervention is superior to another | To determine if a test drug is not inferior to an active control intervention, by a preset margin |
| Null Hypothesis | No difference between the two interventions | The test drug (T) is inferior to the active control (C) by some margin (M) or more (C – T >= M). |
| Alternative Hypothesis | One intervention is superior to the other | The test drug (T) is inferior to the active control (C) by less than M (C-T < M) |
Selecting a non-inferiority margin in a trial is challenging but also critical to a successful trial. The largest possible choice for the non-inferiority margin is the entire known effect of the active control compared to placebo, called M1. However, doing this, would lead to a finding that the test drug has an effect greater than 0. More generally, the non-inferiority margin is set to some portion of M1, called M¬2, to preserve some effect of the control drug, based on clinical judgment. For example, if a superiority trial of the active control demonstrated to be 15% better than placebo, a clinician may set the non-inferiority margin to be 9% (M1=15%, M2=9%). This would be 6% worse than the active treatment, but still 9% better than placebo.
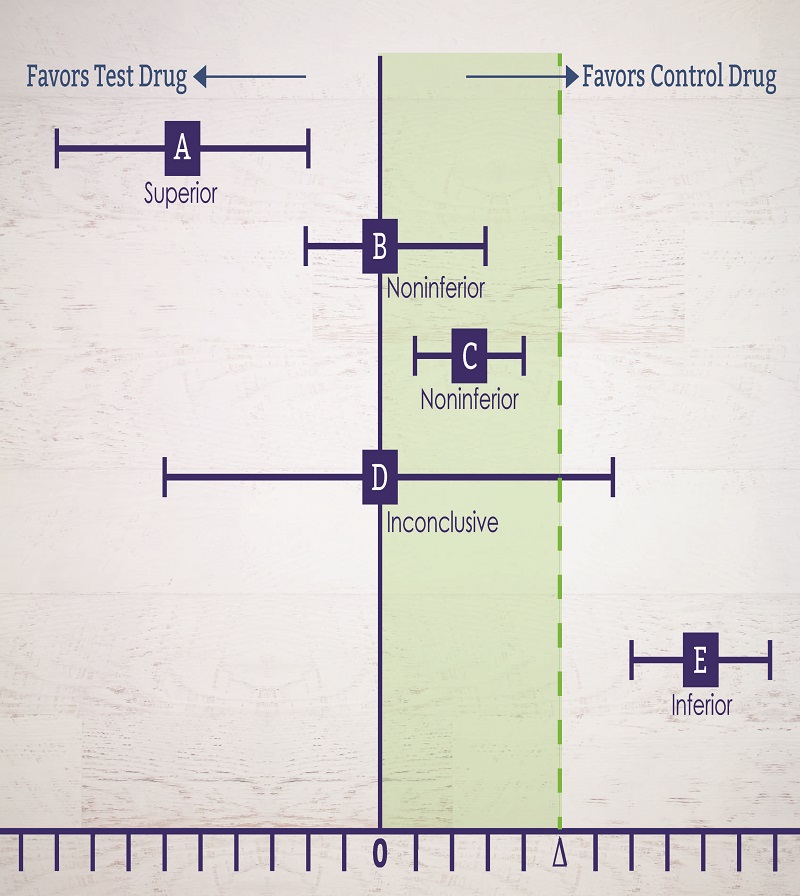
Multiple results are possible in a non-inferiority trial as explained in the graphic below. The point estimate is indicated by the square and is the measure of C – T; the bars represent a 95% confidence interval; and ∆ is the non-inferiority margin.
- Point estimate favors test drug and both superiority and non-inferiority are demonstrated.
- Point estimate is 0 suggesting equal effect of active control and active treatment. The upper bound of the 95% confidence interval is below the non-inferiority margin so non-inferiority is demonstrated.
- The point estimate favors the active control. The upper bound of the 95% confidence interval is less than the non-inferiority margin, demonstrating non-inferiority. However, the point estimate is above zero indicating that active treatment is not as good as the active control (C – T > 0), even while meeting the non-inferiority standard.
- Point estimate is 0 suggesting equal effect, but the upper bound of the 95% confidence interval is greater than the non-inferiority margin so non-inferiority is not demonstrated.
- Point estimate favors the active control and the entire confidence interval is above the non-inferiority margin so inferiority is demonstrated.
Non-inferiority Margin
The selection of the non-inferiority margin is critical in designing a non-inferiority trial and the majority of the FDA guidance focuses on this. The non-inferiority margin is selected by reviewing historical trials of the active control. The active control must be a well-established intervention with at least one superiority trial establishing benefit over placebo. If approval of the active control was based on a single study (not unusual in the setting of risk reduction of major events such as death, stroke, and heart attack), changes in practice should be evaluated. Using the lower bound of the 95% confidence interval provides a conservative estimate of the active control effect. If multiple historical trials exist one of the assumptions of the non-inferiority trial is consistency of the effect between the historical studies and the non-inferiority trial. Therefore, if consistency isn’t present between the historical studies this can lead to problems in estimating the active control effect. Inconsistency can also sometimes lead researchers away from performing a non-inferiority trial, especially if a historical trial did not demonstrate an effect. In situations with multiple historical trials, careful review of all study results and a robust meta-analysis are crucial to selecting an appropriate non-inferiority margin.
Assay Sensitivity and Constancy Assumption
Assay sensitivity is essential to non-inferiority trials as it demonstrates that had the study included a placebo arm, the active control – placebo difference would have been at least M1. The guidance outlines three considerations when determining if a trial has assay sensitivity.
- Historical evidence of sensitivity to drug effects
- The similarity of the new non-inferiority trial to the historical trials (the constancy assumption)
- The quality of the new trial (ruling out defects that would tend to minimize differences between treatments)
The constancy assumption in #2 above is that the non-inferiority study is sufficiently similar to the past studies with respect to the following design features.
- The characteristics of the patient population
- Important concomitant medications
- Definitions and ascertainment of study endpoints
- Dose of active control
- Entry criteria
- Analytic approaches
The presence of constancy is important to evaluate. For example, if a disease definition has changed over time or the methodology used in the historical trial is outdated the constancy assumption may be violated and the use of a non-inferiority design may not be appropriate. If all the design features are similar except the patient characteristics the estimate of the size of the control effect can be adjusted if the effect size is known in the patient sub-groups.
Benefits of non-inferiority trials
- A non-inferiority trial is useful when a placebo controlled trial is not appropriate.
- A non-inferiority trial may also test for superiority without concern about inflating the Type I error rate with care planning of the order in which hypothesis are tested. The reverse is not true; a superiority trial cannot claim non-inferiority.
Disadvantages of non-inferiority trials
- Must be able to demonstrate assay sensitivity and the constancy assumption hold. This is especially difficult when medical practice has changed since the superiority trial (e.g. the active control is always used with additional drugs currently).
- When the active treatment is not well established or historical trials have shown inconsistent results choosing a non-inferiority margin proves to be difficult.
- If the treatment effect of the active control is small, the sample size required for a non-inferiority study may not be feasible