Clinical Trials
Oncology
She thought cancer would prevent her from becoming a mother.
This year, her daughter turns three.
That is what drives us. Experience, Rho.


Cancer is the number two cause of death in both the US and EU. New cancer cases are estimated to increase to 28.4 million by 2040¹, and behind every single one of those numbers is a person. The development of new drugs and treatment regimens has never been more critical—or attainable—as oncology clinical research continues to accelerate. Dramatically more active therapies, and even cures, are now within reach.
Oncology clinical trials can be functionally demanding—and biologically and medically complex—but Rho is up to the challenge.
With on-staff experts from National Cancer Institute (NCI), designated Cancer Centers, years of experience with cancer clinical trials, and more than 35 oncology indications in 28 countries across the US and Europe, we understand clinical trials at all stages—and what it takes to carve out solutions and ensure success.
Rho is prepared to partner for success, and to bring an experienced, stable, and accountable research team to bear in the ongoing battles against cancer.
- Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries [published online ahead of print, 2021 Feb 4]. CA Cancer J Clin. 2021;10.3322/caac.21660.
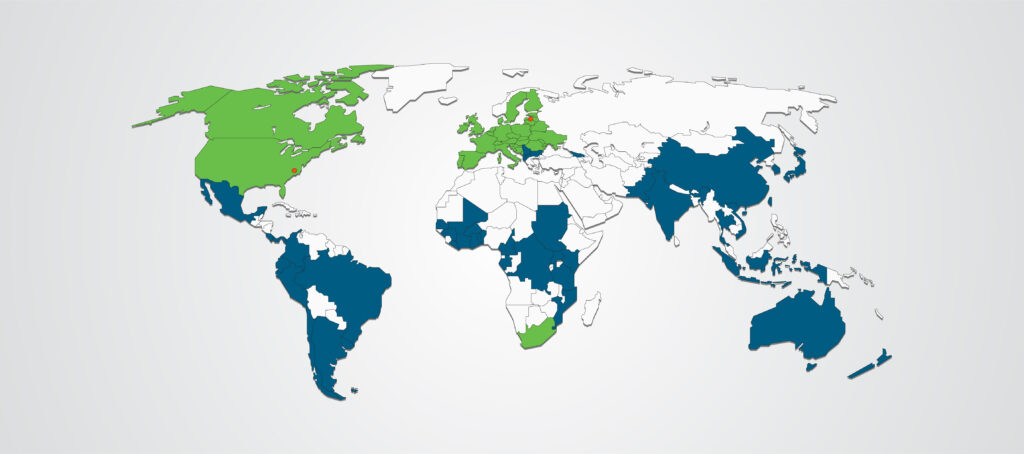
Global Reach
Our global client solutions will ensure you reach your patients, wherever they are. With international capabilities that span 5 continents, we provide full-service capabilities to execute your clinical development programs and trials across the globe.
Together, we can deliver innovative therapies to cancer patients as early as possible, improving their quality of life and offering hope for the future. We proudly invite you to experience Rho.

Our Expertise
10
marketing applications in oncology indications
35+
indications
28
countries
Indications Expertise
-
Solid Tumors
- Advanced metastatic solid tumors
- Anal cancer
- Basal and squamous cell carcinoma
- Bladder cancer
- Brain cancer (primary and metastatic)
- Breast cancer
- Carcinoid tumors
- Cholangiocarcinoma
- Colorectal cancer
- Esophageal cancer
- Gall bladder cancer
- Gastric cancer
- GI stromal tumor
- Glioblastoma multiforme
- Head & neck cancer
- Hepatocellular carcinoma
- Liver cancer
- Lung cancer (NSCLC, SCLC)
- Melanoma
- Myelofibrosis
- Neuroectodermal tumors
- Neuroendocrine
- Ovarian cancer
- Pancreatic cancer
- Pediatric oncology
- Prostate cancer
- Renal cell cancer
- Salivary gland cancer
- Thyroid cancer
-
Hematologic Malignancies
- Acute lymphoblastic leukemia
- Acute myeloid leukemia
- Chronic lymphocytic leukemia
- Chronic myeloid leukemia
- Diffuse large b-cell lymphoma
- Hodgkin’s disease
- Leukemia
- Multiple myeloma
- Non-Hodgkin’s lymphoma
Overcoming the Challenges of Oncology Clinical Trials
Cancer clinical trials can be complex, lengthy, and costly. Site selection, regulatory setup, and data collection all pose challenges, but patient recruitment and retention is one of the biggest hurdles to conducting oncology clinical trials.¹ Less than 5% of adult patients diagnosed with cancer are enrolled in clinical trials², and in situations where eligible patients are limited, trials may compete for study participants.
Rho understands these challenges and can help you achieve your goals, whether you’re facing complicated study designs and treatment regimens or trying to navigate the approval process for a novel therapy with FDA or other regulatory agencies.
Trial Design
Effectively planning a clinical trial can streamline initiation, save resources, and help achieve enrollment goals—which could get new treatments to cancer patients sooner. Rho’s experts can help you:
- Strategize and identify an optimum clinical trial plan and protocol to help ensure successful execution.
- Advise on the best method for data collection and assist with identification and recruitment of trial subjects.
- Explore innovative designs, such as adaptive trials, to maximize learning from each study population.
Trial Strategy and Enrollment
Rho’s experts have extensive experience conducting feasibility studies and ensuring on-time enrollment both in the US and Europe. They can help you:
- Identify and select high-enrolling and quality-driven clinical sites with proven track records.
- Create and implement patient-centric strategies to increase recruitment and retention.
- Ensure medical staff are highly experienced and trained to manage patients with complicated medical histories and well versed in protocol and study procedures.
- Ensure CRAs are well-trained in oncology-specific challenges to support thorough and efficient data collection and review, as well as excellent communication with sites for on-time site start-up and database lock.
Program Strategy and Marketing Applications
Rho’s regulatory strategy and submissions team are experts at guiding oncology development programs, from preclinical through Phase 2/3, toward approval. They have experience navigating FDA’s accelerated approval pathways for clinical development of oncology-related products and have interacted with FDA and other regulatory bodies on behalf of our sponsors. Their understanding of the regulatory framework ensures that your development program will stay on track in this complex therapeutic area. In addition to strategic regulatory consulting, our in-house team of regulatory submissions experts has supported more than 10 marketing application submissions for oncology products and maintains a 100% technical acceptance rate.
- Busting Patient Recruitment Bottlenecks in Oncology Trials. Applied Clinical Trials. 2020. Available at: Busting Patient Recruitment Bottlenecks in Oncology Trials. Accessed on: 28 April 2021.
- Unger JM, Cook E, Tai E, Bleyer A. The Role of Clinical Trial Participation in Cancer Research: Barriers, Evidence, and Strategies. Am Soc Clin Oncol Educ Book. 2016;35:185-198.
Featured Content
Oncology Services
-
Clinical Project Management
-
Clinical Operations Monitoring
-
Global Reach
-
Phase I Clinical Development Services
-
Risk-Based Quality Management
-
Medical Monitoring
-
Data Management
-
Biostatistics
-
Statistical Programming
-
Pharmacovigilance
-
Regulatory Operations
-
Medical Writing
-
Data Standards
-
Quality Assurance





